|
GMP Recombinant Human Interleukin-4
(rHuIL-4)
Description:
Recombinant Human IL-4 produced in E.Coli is a single, non-glycosylated
polypeptide chain containing 130 amino acids and having a molecular mass of
15000 Dalton. The rHuIL-4 is purified by proprietary chromatographic techniques.
Molecular weight: 15 KDa
Identity: rh IL-4 as measured by Elisa and Western Blot
Specific activity: > 13.10 6 units/mg compared to NIBSC standard .
CT-h4.S cell proliferation assay.
650 000 IU /vial
Purity: > 98% as determined by SDS-PAGE and HPLC
Endotoxin content: < 0.1 EU/ µg of IL-4 (LAL test)
Host DNA: < 10 pg/ng protein
Sterility: sterile according to EP Test 2.6.1.
Stabiliser: contains Trehalose ; no preservative.
Physical state: freeze-dried
Stability: 12 months at – 20°C to – 80°C
3 months after reconstitution with water for injection ( as defined in EP
Monograph 0169) when stored at -80°C
Reconstitution: use 500 µL water for injection in class A environment in
order to keep the GMP grade .
Packaging unit: 50 µg protein (Lowry test)
GENTAUR rh IL-4 is manufactured in full compliance with cGMP in facilities
approved by the Belgian Ministry of Health for the production and storage of
medicinal products.
Use:
GENTAUR rh IL-4 is not an approved medicinal product and cannot be
injected as such to patients.
FOR EX VIVO CELL CULTURE AND FOR IN VITRO RESEARCH USE ONLY
Cytokines with high activity for dendritic cell research:
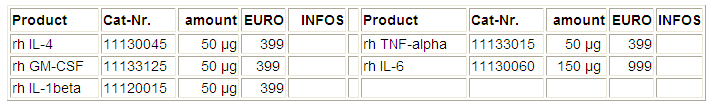
rh: recombinant – human, rm: recombinant - mouse, Net-prices in EURO,
for research only!
________________________________________________________________________________________________________________________
| GMP Recombinant Human Interleukin-4 |
rHuIL-4 |
50 µg |
399 Euro |
|